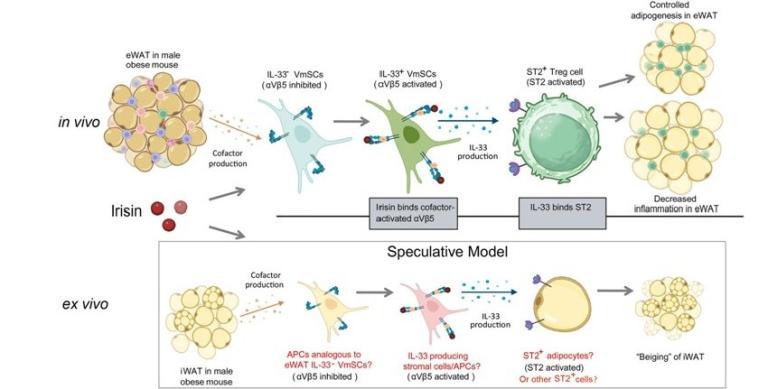
The Spiegelman lab first reported irisin as an exercise-induced myokine in 2012, focusing on its ability to promote "browning" of white adipose tissue. Since then, there has been an explosion of publications on irisin and irisin-mediated effects on diabetes in a variety of animal models and tissues, primarily in the adipose tissue. In the absence of mechanistic studies at the tissue, cellular and molecular levels, it’s been wrongly accepted that irisin acts directly on adipocytes and elicits thermogenesis, accounting for the totality of irisin-mediated improvements in glucose intolerance and insulin resistance. Having first discovered irisin, we feel responsible for identifying the exact pathways at play and their link to the metabolic changes. In this report, we wish to highlight 1) the previously characterized metabolic effects induced by irisin in fat are mediated by the cytokine IL-33 produced by stromal cells, not through a direct interaction between irisin and adipocytes, and 2) irisin-mediated metabolic benefits are largely through ST2+ Treg cells downstream of the induced IL-33 in visceral adipose tissue, while irisin-stimulated thermogenesis of subcutaneous adipose tissue accounts for a smaller fraction of the metabolic changes, which also require IL-33. Therefore, irisin modulates both adipose-tissue inflammation and adipose-tissue thermogenesis through IL-33 that elicits distinct downstream events in different fat depots. It is also important to point out that the reverse of these effects occur in the FNDC5/irisin KO mice, indicating that the effects we described are physiological, not just pharmacological. Additionally, irisin induces weight loss without affecting lean mass, providing a therapeutic pathway alternative to GLP1-based approaches.