Exciting progress has been made in the external sensory systems of vision, somatosensation, olfaction, audition, and gustation, each of which reveals coding strategies that are similar in some regards but unique in others. By contrast, little is known about how the brain monitors the status of internal organs and generates internal senses. The internal sensory system faces unique coding challenges: each organ interprets diverse mechanical, chemical, osmotic, and thermal signals, whereas the same modality in different organs (pressure in the stomach, bladder, lung, and vasculature) can generate categorically distinct responses. Our lack of knowledge on this critically important system prevents a complete understanding of how the brain encodes sensory information and how internal states influence animal behavior.
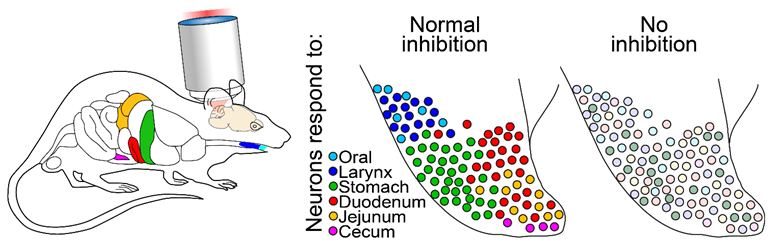
In this study, Chen Ran, Jack Boettcher, Judith Kaye, Catherine Gallori, and Stephen Liberles developed a novel in vivo two-photon mouse brainstem calcium imaging preparation and systems to deliver precisely controlled interoceptive stimuli in the internal organs. They discovered that different organs are represented by discrete and selectively tuned neuronal ensembles, each comprised of heterogeneous molecularly defined cell types. By contrast, neurons responding to distinct mechanical and chemical sensory modalities in the same organ are allowed to converge onto overlapping neuronal populations. Organ representations are topographically organized, forming a “visceral homunculus” in the brainstem. Inhibition is critical for the spatial organization, as blockade of brainstem inhibition broadens neural tuning and blurs sensory representations (Fig. 1). This work provides the first comprehensive characterization of the representations of visceral senses in the brain and reveals basic coding principles used by the brain’s interoceptive system.